Our medical device development experience is defined by our unique blend of advanced engineering, low-volume production expertise, and dedicated support for established medical device companies. As a medical device development company our team can integrate seamlessly with your quality system, offering robust technology and concept development that fast-tracks the path from prototype to production.
From early medical device design through late-stage medical device development, we focus on building reliable, manufacturable solutions that align with regulatory and quality requirements. Our medical device product development capabilities span feasibility, prototyping, verification, and transfer to low-volume production, giving you a single partner to guide your device from concept to commercialization.
We’re currently pursuing our ISO 13485 certification for our production space. Reach out if you have any questions!
We engineer diagnostic and therapeutic tools for rapid, on-site use, supporting medical device development programs that demand portability, usability, and accuracy at the point of care.
We design and develop a range of custom surgical tools and instruments to be used during complex procedures. Our medical device design expertise ensures ergonomic, intuitive tools that integrate seamlessly into the surgical workflow.
We engineer respiratory support devices, including ventilators, oxygen concentrators, monitors, and patient-assistance tools. These medical device development efforts focus on safety, reliability, and performance under demanding clinical conditions.
We develop medical devices for at-home sample collection and preparation, combining user-centered medical device design with robust engineering to support accurate, repeatable results outside the clinical setting.
We use rapid prototyping and design for manufacturing to create implantable devices. As a medical device development company, we collaborate closely with your team to ensure the implant’s form, fit, and function are optimized for both patients and clinicians.
We design and develop devices for diagnosing a wide range of medical conditions. Our medical device product development approach integrates hardware, software, and consumables to deliver complete diagnostic solutions.
Medical Device Expertise
We thrive on the “fuzzy front end” of medical device innovation, where our team embraces open-ended challenges to test the feasibility of new ideas. Our passion is problem-solving, and we excel at assessing new technologies and prototyping to determine if a concept can be made a reality, providing a rapid and flexible approach to early-stage medical device development.
Whether you are refining an existing concept or starting from a blank slate, our medical device design and engineering teams collaborate to de-risk critical functions early. This front-end focus lays a strong foundation for downstream medical device product development, helping you move efficiently toward verification, validation, and production.


We have a certified BSL-2A Biosafety Lab and ISO Class 7 Clean Rooms right here at Re:Build Fikst. While our low-volume production projects will be covered by this certification, our engineering services typically operate under the client’s existing quality system. This approach ensures documentation and risk management remain client-led, allowing our team to stay focused on what we do best: implementing ideas and building devices.
By combining these facilities with our medical device development expertise, we support activities ranging from early feasibility builds to controlled low-volume production. This integrated environment enables smoother transitions between design iterations, testing, and final medical device product development.
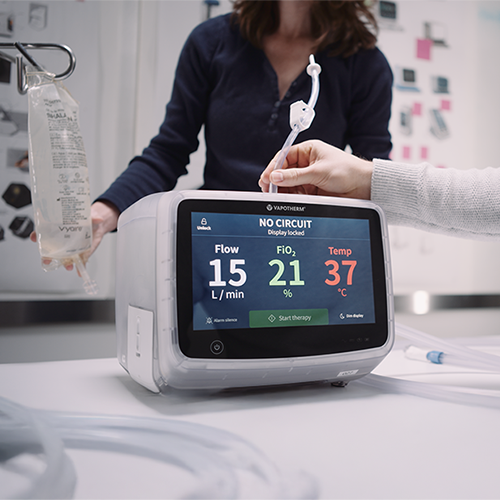
Vapotherm partnered with Re:Build Fikst to develop the HVT 2.0, a next-generation respiratory therapy device. The goal was to create a highly reliable, precise device to treat complex respiratory conditions and reduce the need for invasive procedures like endotracheal intubation. The project also included the development of a disposable patient circuit for clinical use.
If you’re looking for a partner in medical device development, we’re ready to help. Fill out the form below and someone from our team will be in touch with you shortly to discuss your medical device design and development needs.